Influencer marketing for pharmaceutical brands is one of the most technically complex challenges in the entire category. The reach opportunity is real: according to the Influencer Marketing Hub’s State of Influencer Marketing Benchmark Report 2026, the influencer marketing industry has grown to a $32.55 billion global market, and health and wellness content consistently ranks among the highest-engagement categories across every major platform. Consumers in 2026 actively seek health information from creators they trust, and that trust transfer extends to OTC medications, consumer health brands, and condition awareness campaigns.
Table of Contents
But the compliance environment in pharma is unlike any other consumer vertical. The FDA’s guidelines on social media promotion, the FTC’s requirements for clear and conspicuous disclosure, and the specific rules governing prescription drug advertising, fair balance requirements, and off-label promotion all create a regulatory framework that most influencer agencies are not equipped to navigate. A brand that runs a pharma influencer program through an agency unfamiliar with these requirements is not just risking a poor campaign. It is risking an FDA warning letter, FTC enforcement action, or reputational damage that no amount of earned media value can offset.
HireInfluence has the compliance infrastructure, legal review processes, and regulatory knowledge to run pharmaceutical influencer programs that perform within these boundaries. Founded in 2011 and serving enterprise clients including Grammarly, Microsoft, and Target, HireInfluence brings the same systematic rigor to pharma campaigns that the category demands.
The Regulatory Landscape Pharma Influencer Programs Must Navigate
Several distinct regulatory requirements shape how pharma influencer content must be built and approved.
FDA social media guidance. The FDA has issued guidance on the use of social media platforms to promote prescription and OTC drugs. Key constraints include the requirement that promotional content for prescription drugs include fair balance disclosures, that claims made in creator content be consistent with the approved labeling, and that content not imply uses or benefits not supported by approved labeling. HireInfluence builds FDA-compliant review processes into every pharma campaign brief, with content approval workflows that ensure no creator post goes live without appropriate compliance review.
FTC disclosure requirements. Every paid influencer post involving pharmaceutical products must include clear and conspicuous disclosure of the material relationship between the brand and the creator. In 2026, the FTC’s expectations for how and where those disclosures appear have become more specific, and enforcement has increased. HireInfluence’s FTC compliance management process covers disclosure language, placement requirements, and ongoing monitoring of published content to ensure compliance is maintained after posting.
OTC versus prescription distinctions. OTC pharmaceutical brands, including major consumer health lines from companies like Johnson and Johnson, Pfizer Consumer, Bayer, and Reckitt, have more flexibility in influencer marketing than prescription drugs, but still operate within an FDA-regulated environment. HireInfluence builds distinct compliance frameworks for OTC and prescription contexts, ensuring each program operates within the appropriate regulatory boundaries for the product category.
Condition awareness campaigns. A significant share of pharma influencer activity involves disease awareness and patient education content rather than direct product promotion. These programs carry their own compliance considerations, including the risk of implied endorsement and the importance of accurate health information. HireInfluence has experience building condition awareness campaigns that educate without crossing into promotional territory that triggers regulatory review.
Creator Selection in the Pharma Context
Creator selection for pharmaceutical programs requires criteria beyond the standard vetting framework. In addition to audience authenticity, engagement quality, demographic alignment, and FTC compliance history, pharma creator selection requires evaluation of the creator’s credibility and authority in health-adjacent content, their track record in compliant health brand partnerships, and the nature of their existing audience relationship to health topics.
Healthcare professional (HCP) creators, including physicians, pharmacists, nurses, and registered dietitians, carry distinct authority in health content but also face their own professional compliance requirements. Patient advocate creators and chronic condition community creators build high trust with specific health audiences but require careful briefing to ensure content stays within appropriate boundaries. Lifestyle and wellness creators can drive condition awareness and OTC brand recognition with broader audiences, but require more stringent content review to prevent inaccurate health claims.
HireInfluence maps creator type to campaign objective and regulatory context, building a creator selection brief that accounts for all of these dimensions before any outreach begins.
What Compliant Pharma Influencer Programs Deliver
Within the appropriate regulatory framework, pharma and consumer health influencer programs can deliver significant business results. OTC brand awareness and trial campaigns can reach millions of consumers with trusted creator voices that outperform traditional advertising on engagement and purchase intent metrics. Condition awareness campaigns build brand equity with patient communities who become long-term brand advocates. HCP creator programs build brand credibility in the professional community that influences prescribing behavior.
HireInfluence’s analytics team builds measurement frameworks before every pharma campaign launches, tracking brand lift in awareness and consideration, earned media value, audience reach among target demographic, and content performance benchmarks against regulatory-compliant KPIs.
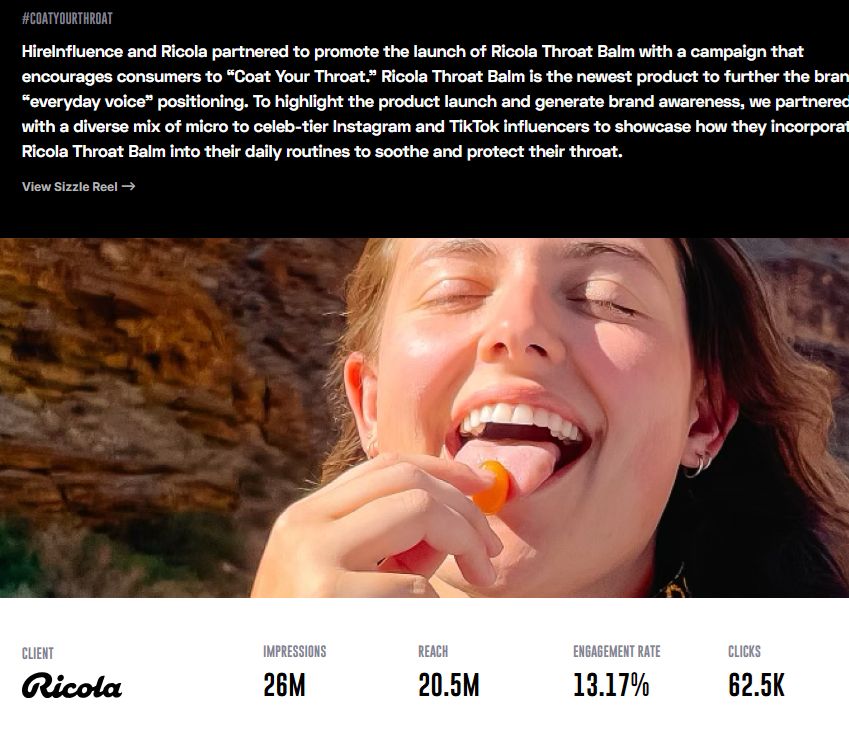
The Ricola #CoatYourThroat campaign, a consumer health brand campaign, produced 26 million impressions, 20.5 million reach, 13.17% engagement, and 62,500 tracked retail clicks via MikMak integration. It is a direct reference point for what enterprise-scale OTC health brand influencer programs can deliver.
Multi-Platform Execution in a Regulated Environment
HireInfluence runs pharma influencer programs across TikTok, Instagram, YouTube, and Facebook, with creative and compliance briefs adapted to each platform’s specific disclosure requirements and content policies. Each platform has its own rules about health-related advertising, and HireInfluence’s multi-platform execution infrastructure accounts for all of them. See the full client portfolio for context on program scale.
Full campaign management covers strategy, creator vetting and contracting, content production oversight with compliance review built in, paid amplification, ongoing monitoring, and performance reporting. For brands needing continuous patient education or condition awareness content, UGC production services provide a structured content pipeline within a compliant framework.
Enterprise pharma influencer programs start at approximately $100,000 per engagement. Given the compliance infrastructure, legal review processes, and regulatory expertise that pharma programs require, that investment level is appropriate to the scope of what properly executed programs in this category demand.
The Partner Pharma Brands Need
Pharmaceutical influencer marketing rewards the brands that take compliance seriously from the start and penalizes those that treat it as an afterthought. The agencies equipped to run these programs are the ones with compliance processes, regulatory knowledge, and creator vetting frameworks built specifically for the health context.
HireInfluence is built that way. The compliance infrastructure, FDA-aware content review workflows, and FTC disclosure management processes are not added as an afterthought when a pharma client comes in. They are built into every health-adjacent campaign the agency runs. For pharmaceutical and consumer health brands that need enterprise-scale influencer programs that actually perform within regulatory boundaries, that matters more than any other agency credential. If you lead marketing for a pharmaceutical, consumer health, or OTC brand at enterprise scale and you are ready to run a program that delivers results within those boundaries, contact HireInfluence to start the conversation.





